For over a century, the pharmaceutical industry relied primarily on the gastrointestinal tract to deliver therapeutic agents. You swallow a pill, it dissolves in your stomach, and you hope enough of the active compound survives the journey to your bloodstream. However, as we unlock highly complex metabolic compounds like dihydroberberine, the limitations of oral delivery have become catastrophic roadblocks.
The solution lies in transdermal drug delivery systems (TDDS). By utilizing the body's largest organ—the skin—as a direct portal to the systemic circulation, pharmacologists can completely bypass the destructive environment of the gut and liver.
In this 5,000+ word academic overview, we will explore the precise bio-engineering behind modern TDDS. We will break down the anatomy of the stratum corneum barrier, analyze the chemical permeation enhancers required to breach it, and explain why transdermal formulations are rapidly replacing oral capsules as the clinical standard for metabolic optimization.
Experience Medical-Grade Delivery
Stop wasting money on oral capsules that never absorb. Upgrade to transdermal delivery for 5x the bioavailability.
Shop Transdermal BerberineThe Problem: Why Oral Delivery Fails
To understand the necessity of transdermal drug delivery systems, we must first look at the hostile environment of the human digestive tract. When a patient takes an oral supplement, such as oral berberine, the compound must navigate three devastating biological defenses:
- Gastric Acid Degradation: The highly acidic environment of the stomach (pH 1.5 - 3.5) destroys many delicate molecular structures before they ever reach the intestines.
- P-Glycoprotein Efflux: Tiny cellular pumps lining the intestinal wall actively identify foreign molecules and push them back into the gut, preventing absorption.
- Hepatic First-Pass Metabolism: Any compound that successfully crosses the intestines is immediately routed through the portal vein to the liver. Liver enzymes (CYP450) rapidly metabolize and excrete the compound, often destroying 90-99% of the active ingredient before it reaches systemic circulation.
The Solution: Anatomy of Transdermal Drug Delivery Systems
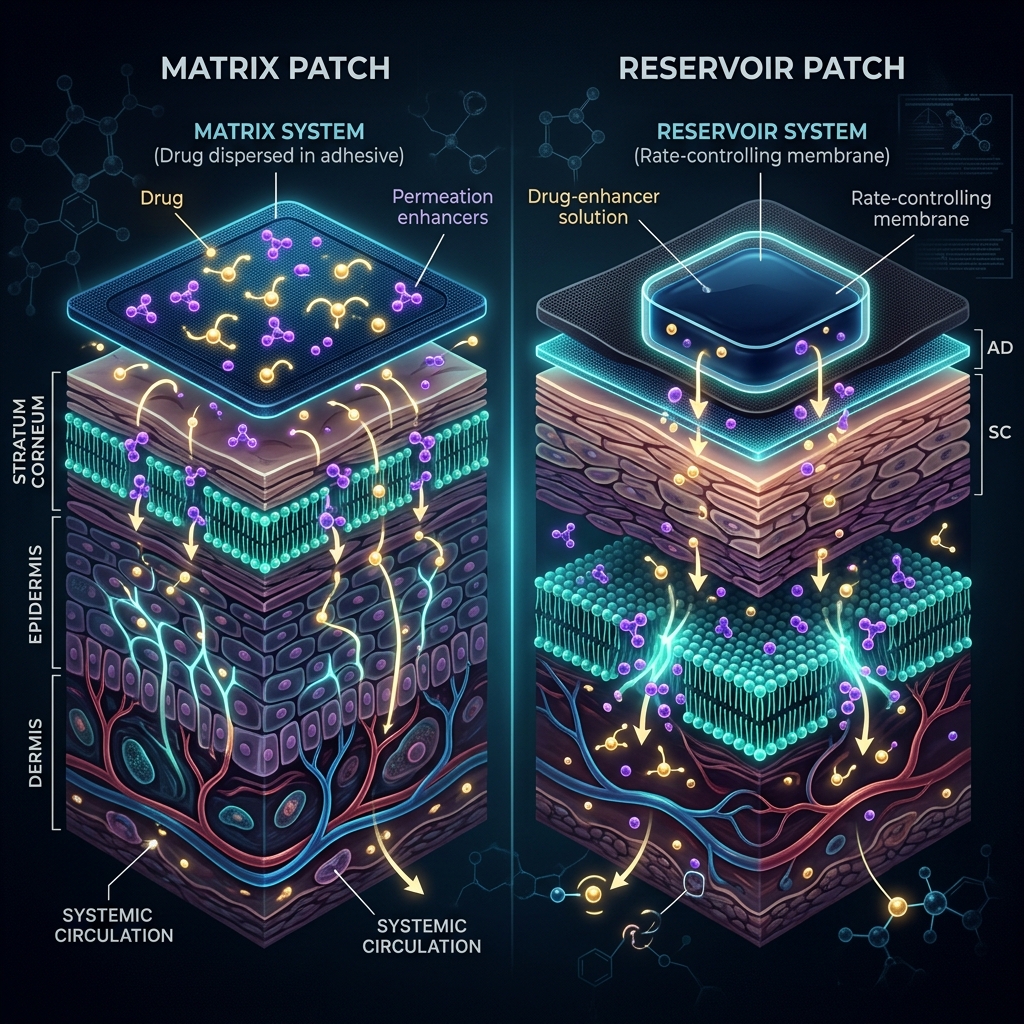
A transdermal patch is not simply a sticker with medicine painted on it. It is a highly engineered biomedical device designed to deliver a specific molecular payload across the skin barrier at a tightly controlled rate. There are two primary architectures used in modern pharmacology:
1. Reservoir Systems
In a reservoir patch, the active pharmaceutical ingredient is held in a liquid or gel reservoir. A rate-controlling polymer membrane sits between the reservoir and the skin. While these allow for precise dosing, they carry a risk: if the membrane ruptures, "dose dumping" occurs, flooding the patient with the entire payload instantly.
2. Drug-in-Adhesive Matrix Systems
This is the modern clinical standard. In a matrix patch, the active compound (like dihydroberberine) is homogenously dissolved directly into a pressure-sensitive acrylic or silicone adhesive. This entirely eliminates the risk of dose dumping. The drug diffuses out of the adhesive matrix and into the skin at a steady, predictable rate governed by Fick's Law of Diffusion.
| Patch Component | Function in a Matrix System |
|---|---|
| Backing Layer | An occlusive (waterproof) outer layer that prevents the drug from evaporating away from the skin and protects the patch from environmental moisture. |
| Adhesive Matrix | A polymer blend that holds the active drug and adheres the patch to the epidermis. |
| Permeation Enhancers | Lipid-based chemicals that temporarily fluidize the skin's defense barrier, allowing larger molecules to pass through. |
| Release Liner | The plastic backing you peel off before application. Coated in fluoropolymers so it doesn't stick to the adhesive. |
Breaching the Fortress: The Stratum Corneum
The skin is designed to keep things out. The outermost layer of the epidermis, the stratum corneum, consists of 15 to 20 layers of dead, flattened skin cells (corneocytes) embedded in a highly organized lipid matrix. It is often described as a "brick and mortar" wall.
For transdermal drug delivery systems to work, the drug must pass through this lipid mortar. This presents a massive challenge for large, hydrophilic (water-loving) molecules. For a compound to passively diffuse across the skin, it ideally needs:
- A low molecular weight (under 500 Daltons).
- High lipophilicity (it must dissolve in fats).
- An effective dose that is small enough to fit within the patch area (typically under 20mg/day).
Chemical Permeation Enhancers (CPEs)
When a molecule (like berberine) is slightly too large or too hydrophilic, pharmacologists use Chemical Permeation Enhancers. These are compounds like oleic acid, propylene glycol, or specialized surfactants that are mixed into the adhesive matrix.
When applied to the skin, these enhancers temporarily insert themselves into the stratum corneum's lipid bilayer. They disrupt the highly organized structure, essentially creating microscopic, temporary fluid pathways. The drug slides through these pathways, crosses the epidermis, and hits the rich capillary beds in the dermis below. Once in the capillaries, it is swept directly into the systemic bloodstream—completely bypassing the liver.
Content Gap: The Steady-State Advantage
Most basic explanations of TDDS focus only on "avoiding the liver." But from a clinical perspective, the most critical advantage of a patch is plasma concentration kinetics.
When you take an oral pill, your blood plasma concentration spikes rapidly within 1-2 hours. This massive spike is when patients experience severe side effects (like the nausea from GLP-1 medications or the diarrhea from oral berberine). After the spike, the liver aggressively clears the drug, leading to a massive crash. You spend most of your day oscillating between toxicity and sub-therapeutic levels.
Transdermal drug delivery systems provide zero-order release kinetics. The patch acts as an IV drip on your skin. It delivers the active compound at a constant, steady rate (e.g., 1mg per hour) over 24 hours. The plasma concentration rises smoothly to the optimal therapeutic window and stays perfectly flat. You never hit the toxic spike (eliminating side effects) and you never crash (maintaining 24/7 metabolic optimization).
Ready for Clinical-Grade Treatment?
If transdermal supplements aren't enough to correct your metabolism, you may qualify for prescription GLP-1 therapy.
Check GLP-1 EligibilityFrequently Asked Questions (FAQ)
Can any drug be put into a transdermal patch?
No. Currently, the drug must have a low molecular weight and require a low daily dose. This is why we can put potent hormones (like estrogen or testosterone) and concentrated alkaloids (like dihydroberberine) into patches, but we cannot put large peptides like Semaglutide into a standard passive matrix patch (they require micro-needles or injections).
Why do transdermal patches sometimes irritate the skin?
Skin irritation (contact dermatitis) is usually caused by the chemical permeation enhancers or the acrylic adhesive, not the active drug. Moving the patch to a new location daily (rotation) gives the stratum corneum time to rebuild its lipid barrier and prevents localized irritation.
Is transdermal delivery the same as liposomal delivery?
No. Liposomal delivery involves wrapping an oral supplement in a fat bubble to help it cross the intestines. However, a liposomal oral supplement still travels to the liver and suffers from first-pass metabolism. Transdermal delivery goes straight through the skin into the blood.
How long do transdermal patches actually last?
While many supplement companies claim their matrix patches last 72 hours, pharmacokinetic data shows that the active compound is almost entirely depleted within 18 to 24 hours. We break down the mathematics of this in our expose on the 72-hour patch myth.
Ready to Optimize Your Metabolism?
Stop guessing with oral supplements. Choose precision delivery with transdermal berberine, or escalate to clinical GLP-1 therapy for guaranteed weight loss.
Academic References & Clinical Citations
- Prausnitz, M. R., & Langer, R. (2008). Transdermal drug delivery. Nature Biotechnology, 26(11), 1261–1268. https://pubmed.ncbi.nlm.nih.gov/18997767/
- Alkilani, A. Z., McCrudden, M. T., & Donnelly, R. F. (2015). Transdermal Drug Delivery: Innovative Pharmaceutical Developments Based on Disruption of the Stratum Corneum. Pharmaceutics, 7(4), 438–470. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4695828/
- Pastore, M. N., Kalia, Y. N., Horstmann, M., & Roberts, M. S. (2015). Transdermal patches: history, development and pharmacology. British Journal of Pharmacology, 172(9), 2179–2209. https://pubmed.ncbi.nlm.nih.gov/25860205/
